ABOUT
Description



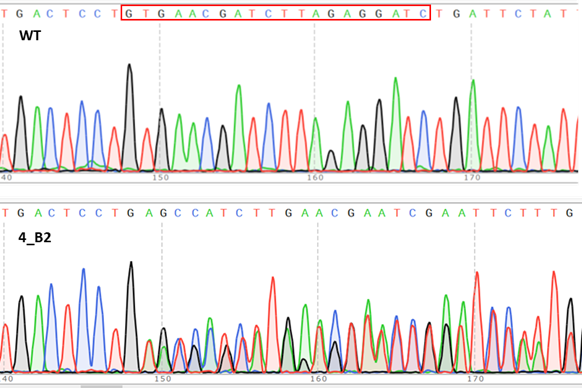
Biomedical research has significantly advanced in the recent years thanks, in part, to the discovery of human induced pluripotent stem cells (hiPSCs), the development of CRISPR/Cas9 gene editing and the possibility of generating specific differentiation systems and more complex in vitro organ-like structures called organoids. These technologies have become key players in investigating disease modelling, embryology and for novel regenerative therapeutic approaches that currently enter first clinical trials.
Core facilities in European research centers and universities providing services in these fields are becoming a reference hub for know-how for these technologies. Furthermore, they establish initial quality control standards and homogenize procedures to enhance reproducibility in biomedical research. However, one of the major challenges for core facilities is to keep track of all advancements in cutting-edge technologies.
New protocols, reagents and tools continuously develop and need to be tested. Isolated technical platforms cannot follow the fast-moving technology pace. Therefore, there is an urgent need to join forces and link these core facilities to harmonize methodologies used to increase the overall reproducibility of the results produced in different labs, from different institutions within Europe and to increase their impact.
CorEuStem is composed of experts in stem cell, differentiation, organoids and gene editing technologies with the aim of joining forces and establishing the first European network for harmonizing procedures and protocols, to organise joint training schools for implementing new cutting-edge technologies emerging in the field and to become a reference point in stem cells, differentiation, organoids and gene editing in Europe and beyond.
ABOUT
Specific
Objectives
The Core objective of the CorEuStem consortium has been to establish a validated set of protocols, procedures and tools to provide efficient benchmarking, quality control and intensive training.
Research Coordination
- RCO1: Implement a benchmarking and quality control methodology across facilities.
- RCO2: Establish CorEuStem annual meetings and workshops with the aim to keep members up to date with the the status of the field/technologies and identifying new trends and new technologies emerging in stem cell biology, organoids, gene editing and their applications.
- RCO3: Establishment and implementation of a high quality training program through the creation of the CorEuStem training school.
Capacity Building
- CBO1: Creation of a centrally accessible web-based platform for discussion, validated reagents, protocols and quality control tests that will regularly be updated by the responsible working groups according to most recent advances.
- CBO2: To foster international cooperation for the development of new strategies, and quality control standards for processes and protocols. Defined working groups with common interests and objectives that will work together.
- CBO3: Establish integrated projects across facilities and across national boundaries.
- CBO4: The Action will facilitate applications to future coordinated funding schemes to further develop new technologies and to enhance research in Europe in the fields of stem cell biology, organoids and gene editing.
- CBO5: The Action will initiate collaborative innovative state of the art technological developments using stem cells, organoids, genetic manipulation and characterization together with research labs in the field and to combine it with screening, microfluidics, single cell technologies, 3D bioprinting, imaging technologies and artificial intelligence.
ABOUT
Challenge
Human PSC technologies have advanced rapidly over the past 20 years. Currently, in Europe, core facilities and laboratories with expertise in these technologies are concentrated in a few countries. It is predicted that in the next few years, most of the European countries will establish laboratories and core facilities focused on hPSC technologies.
One of the major challenges for core facilities is to keep track, evaluate and implement cutting-edge technologies. There is therefore a need to link core facilities and laboratories together to develop consensus methodologies to be adopted as best practice by the stem cell community to increase the overall reproducibility and comparability of outputs from laboratories in Europe.
The network will work towards delivering these objectives. Challenges that Core facilities working with stem cells, organoids and gene editing face and that the network will be able to address are:
- Defining current best practices. Evaluation of different cell culture conditions and procedures to minimize the genetic instability. Identify robust methods to analyse the safety and potency of hPSCs and their differentiated cell types.
- Generation of a comprehensive validated data set. This data set will contain information for a well characterized set of reference iPSCs with associated data optimised for defined culture conditions. The reference iPSC lines are already available throughout the network, with proper consents covering banking, research and intellectual property rights. This data set will include such parameters as optimised differentiation protocols, CRISPR/Cas9 gene editing efficiencies and chromosomal stability over time in culture. As a network, working with large cohorts of different cell lines from both normal and disease backgrounds, the consortium can create working groups testing the reproducibility and robustness of protocols leading to optimization. This will be a valuable and much needed resource for the stem cell field. Again, use of reference cell lines will be a strong asset.
- Harmonize procedures within Europe. Establish the criteria and testing strategies for establishing a robust comparability of datasets between European labs when using gene editing and stem cell technologies. The establishment of quality assessment standards will have a positive effect on the quality and reproducibility of data generated and published throughout Europe.
- Benchmark and continuous training. Enhancing the skillsets of core facilities through training personnel in new technologies emerging in the field, achieved through a dynamic collaborative inter-laboratory training programme. The network will be crucial to disseminate knowledge and current best practice across Europe.
ABOUT
Background
In the past 12 years, experimental approaches used in biomedical research have been reshaped by the discovery of human induced pluripotent stem cells (iPSCs), the development of CRISPR/Cas9 gene editing technology and the possibility of generating in vitro organ-like structures called organoids.
A challenge in the adoption and execution of these complex technologies lies in the Action’s ability to design robust protocols in these areas, establish quality control standards and provide specific training. The iPSC core facilities are at the forefront of these dependencies. The value of iPSC cores comes from their skillsets and purpose:
- cores have to implement, optimize and establish robust protocols and quality control standards that will work on every pluripotent cell line they are required to work with and
- cores have to train their own staff and staff in research labs, often with no prior knowledge for work with pluripotent-stem cell-based projects.